Irb Form - For more information on how to submit through cayuse, please the training manuals. To submit a protocol, please go to cayuse. Find irb review application (ira) forms, guidance documents, and submission templates and checklists here. Our forms and guidance documents are often updated as. Always download fresh forms and templates with each new irb submission. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. For a list of all guidelines, policies, forms, tools, and templates related to the revised common rule click here.
Find irb review application (ira) forms, guidance documents, and submission templates and checklists here. Always download fresh forms and templates with each new irb submission. To submit a protocol, please go to cayuse. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. For more information on how to submit through cayuse, please the training manuals. For a list of all guidelines, policies, forms, tools, and templates related to the revised common rule click here. Our forms and guidance documents are often updated as.
For more information on how to submit through cayuse, please the training manuals. Our forms and guidance documents are often updated as. To submit a protocol, please go to cayuse. For a list of all guidelines, policies, forms, tools, and templates related to the revised common rule click here. Find irb review application (ira) forms, guidance documents, and submission templates and checklists here. Always download fresh forms and templates with each new irb submission. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp.
IRB Application Form
Our forms and guidance documents are often updated as. Always download fresh forms and templates with each new irb submission. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. For more information on how to submit through cayuse, please the training manuals. For a list of all guidelines, policies,.
IRB APPLICATION . Application for human subjects research Doc Template
Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. For a list of all guidelines, policies, forms, tools, and templates related to the revised common rule click here. Always download fresh forms and templates with each new irb submission. For more information on how to submit through cayuse, please.
IRB Application Form PDF
Always download fresh forms and templates with each new irb submission. Our forms and guidance documents are often updated as. To submit a protocol, please go to cayuse. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. Find irb review application (ira) forms, guidance documents, and submission templates and.
SOLUTION Irb Form Studypool
Our forms and guidance documents are often updated as. For a list of all guidelines, policies, forms, tools, and templates related to the revised common rule click here. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. Find irb review application (ira) forms, guidance documents, and submission templates and.
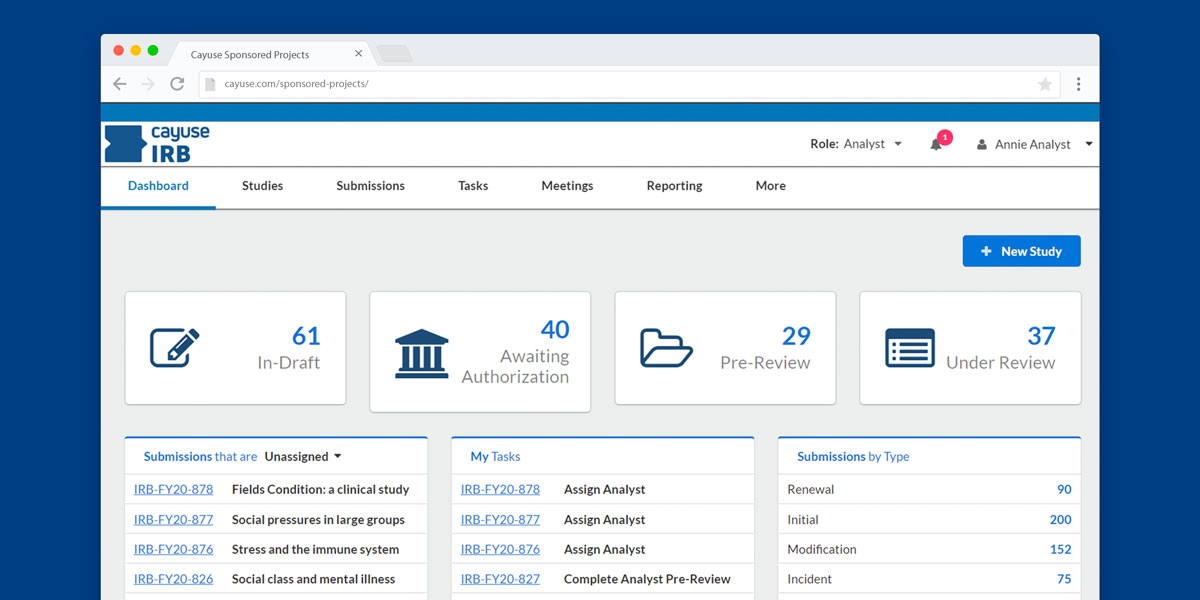
IRB Software for Review & Approval Easy Management & Compliance
To submit a protocol, please go to cayuse. Our forms and guidance documents are often updated as. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. Find irb review application (ira) forms, guidance documents, and submission templates and checklists here. For more information on how to submit through cayuse,.
institutional review board (irb) authorization agreement Doc Template
Our forms and guidance documents are often updated as. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. Always download fresh forms and templates with each new irb submission. To submit a protocol, please go to cayuse. Find irb review application (ira) forms, guidance documents, and submission templates and.
IRB Consent Form Word PDF Google Docs
For more information on how to submit through cayuse, please the training manuals. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. For a list of all guidelines, policies, forms, tools, and templates related to the revised common rule click here. Our forms and guidance documents are often updated.
IRB Consent Template Minimal Risk Studies Doc Template pdfFiller
Our forms and guidance documents are often updated as. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. Always download fresh forms and templates with each new irb submission. To submit a protocol, please go to cayuse. Find irb review application (ira) forms, guidance documents, and submission templates and.
Institutional Review Board Application to Conduct Research Doc
Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. Our forms and guidance documents are often updated as. For a list of all guidelines, policies, forms, tools, and templates related to the revised common rule click here. Find irb review application (ira) forms, guidance documents, and submission templates and.
Authorization Letter Sample download free documents for PDF, Word and
For more information on how to submit through cayuse, please the training manuals. For a list of all guidelines, policies, forms, tools, and templates related to the revised common rule click here. Completion of the irb roster form is required if your irb is designated on a federalwide assurance submitted to ohrp. Always download fresh forms and templates with each.
Completion Of The Irb Roster Form Is Required If Your Irb Is Designated On A Federalwide Assurance Submitted To Ohrp.
Our forms and guidance documents are often updated as. To submit a protocol, please go to cayuse. For more information on how to submit through cayuse, please the training manuals. Find irb review application (ira) forms, guidance documents, and submission templates and checklists here.
Always Download Fresh Forms And Templates With Each New Irb Submission.
For a list of all guidelines, policies, forms, tools, and templates related to the revised common rule click here.