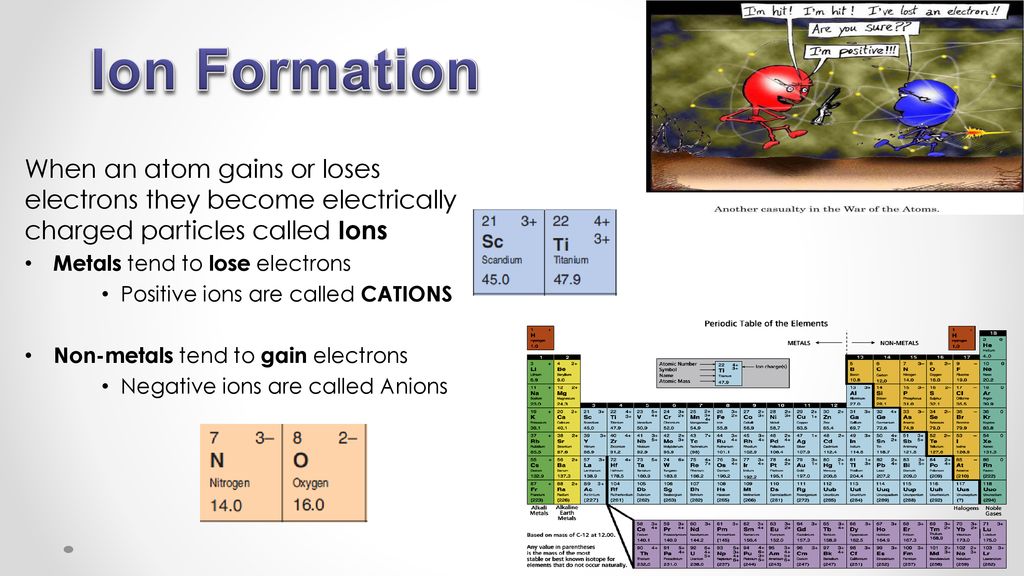
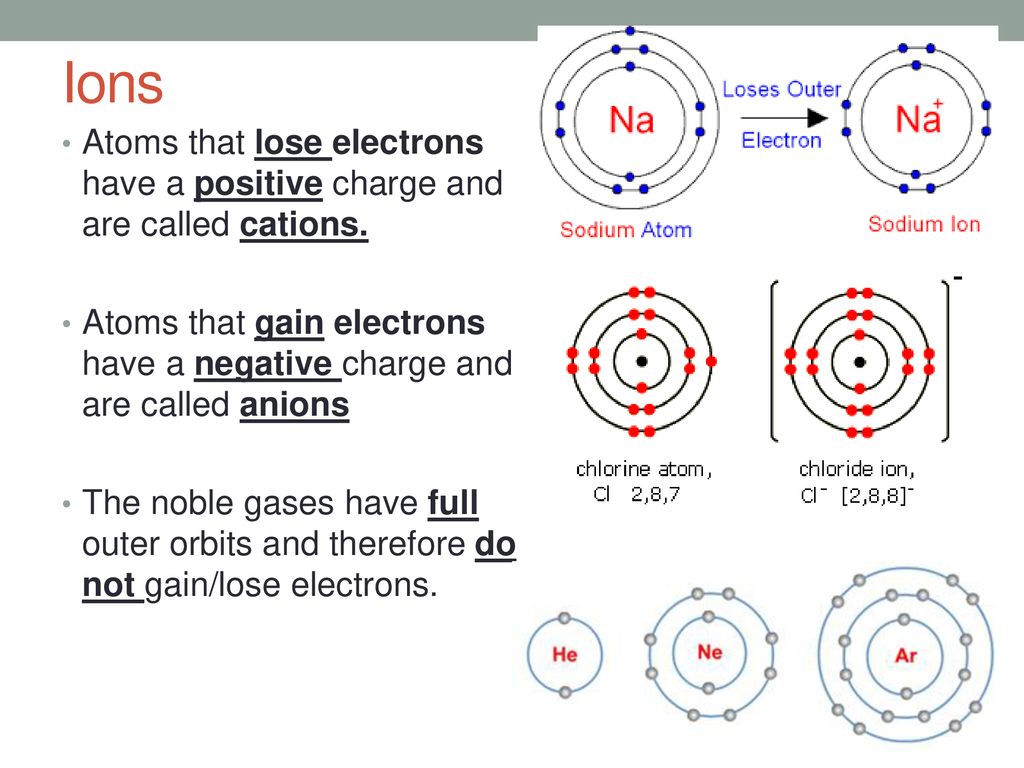
Ions Are Formed When Atoms Gain Or Lose - Ionic bonds occur between atoms from adjacent. Atoms gain or lose electrons in order to attain a valence orbital arrangement like that of a noble gas. Study with quizlet and memorize flashcards containing terms like ions form when., which electrons do atoms gain/lose to form ions?, why. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the positive charges of the. A positive ion is formed when an element, typically a metal loses 1 or more. Describe two ways that an ion forms from an atom. Ions are formed when an atome either loses or gains an electron.
Ionic bonds occur between atoms from adjacent. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the positive charges of the. A positive ion is formed when an element, typically a metal loses 1 or more. Atoms gain or lose electrons in order to attain a valence orbital arrangement like that of a noble gas. Ions are formed when an atome either loses or gains an electron. Describe two ways that an ion forms from an atom. Study with quizlet and memorize flashcards containing terms like ions form when., which electrons do atoms gain/lose to form ions?, why.
Study with quizlet and memorize flashcards containing terms like ions form when., which electrons do atoms gain/lose to form ions?, why. A positive ion is formed when an element, typically a metal loses 1 or more. Describe two ways that an ion forms from an atom. Ionic bonds occur between atoms from adjacent. Atoms gain or lose electrons in order to attain a valence orbital arrangement like that of a noble gas. Ions are formed when an atome either loses or gains an electron. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the positive charges of the.
Atomic Structure and Ions GCSE Chemistry Revision
Describe two ways that an ion forms from an atom. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the positive charges of the. A positive ion is formed when an element, typically a metal loses 1 or more. Study with quizlet and memorize flashcards containing terms like ions form.
Forming compounds day 2 Ionic Bonds ppt download
Ions are formed when an atome either loses or gains an electron. Study with quizlet and memorize flashcards containing terms like ions form when., which electrons do atoms gain/lose to form ions?, why. Atoms gain or lose electrons in order to attain a valence orbital arrangement like that of a noble gas. Atoms that lose electrons acquire a positive charge.
Forming Ions GCSE Chemistry Revision
Study with quizlet and memorize flashcards containing terms like ions form when., which electrons do atoms gain/lose to form ions?, why. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the positive charges of the. Atoms gain or lose electrons in order to attain a valence orbital arrangement like that.
Ion Formation When an atom gains or loses electrons they
Describe two ways that an ion forms from an atom. Study with quizlet and memorize flashcards containing terms like ions form when., which electrons do atoms gain/lose to form ions?, why. Atoms gain or lose electrons in order to attain a valence orbital arrangement like that of a noble gas. Atoms that lose electrons acquire a positive charge because they.
Ions and Ionic Compounds ppt download
Describe two ways that an ion forms from an atom. Study with quizlet and memorize flashcards containing terms like ions form when., which electrons do atoms gain/lose to form ions?, why. Ionic bonds occur between atoms from adjacent. A positive ion is formed when an element, typically a metal loses 1 or more. Atoms that lose electrons acquire a positive.
The Chemistry of Life Day ppt download
Study with quizlet and memorize flashcards containing terms like ions form when., which electrons do atoms gain/lose to form ions?, why. Ionic bonds occur between atoms from adjacent. A positive ion is formed when an element, typically a metal loses 1 or more. Describe two ways that an ion forms from an atom. Ions are formed when an atome either.
Unit 5 Ionic Bonding & Nomenclature ppt download
Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the positive charges of the. A positive ion is formed when an element, typically a metal loses 1 or more. Ions are formed when an atome either loses or gains an electron. Describe two ways that an ion forms from an.
Ionic Compounds and Metals ppt download
Ionic bonds occur between atoms from adjacent. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the positive charges of the. Study with quizlet and memorize flashcards containing terms like ions form when., which electrons do atoms gain/lose to form ions?, why. Atoms gain or lose electrons in order to.
Chapter 19 Molecules and Compounds ppt download
Describe two ways that an ion forms from an atom. Atoms gain or lose electrons in order to attain a valence orbital arrangement like that of a noble gas. Ions are formed when an atome either loses or gains an electron. Ionic bonds occur between atoms from adjacent. A positive ion is formed when an element, typically a metal loses.
Notes Ionic Bonds 1. Key Concept Ionic bonds form when electrons are
Ionic bonds occur between atoms from adjacent. Describe two ways that an ion forms from an atom. Ions are formed when an atome either loses or gains an electron. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the positive charges of the. A positive ion is formed when an.
Describe Two Ways That An Ion Forms From An Atom.
Ions are formed when an atome either loses or gains an electron. Atoms gain or lose electrons in order to attain a valence orbital arrangement like that of a noble gas. A positive ion is formed when an element, typically a metal loses 1 or more. Atoms that lose electrons acquire a positive charge because they are left with fewer negatively charged electrons to balance the positive charges of the.
Study With Quizlet And Memorize Flashcards Containing Terms Like Ions Form When., Which Electrons Do Atoms Gain/Lose To Form Ions?, Why.
Ionic bonds occur between atoms from adjacent.