How Many Covalent Bonds Does Oxygen Form - If sulfur has an atomic number of 16 how many covalent bonds does it form? Sulfur can form up to two covalent bonds because it has six. Carbon and hydrogen do not typically form hydrogen bonds. A hydrogen atom can form a maximum of one covalent bond. The sulfur atom has a total of 6 valence electrons, with two bonds to oxygen atoms and two bonds to chlorine atoms.
Sulfur can form up to two covalent bonds because it has six. If sulfur has an atomic number of 16 how many covalent bonds does it form? The sulfur atom has a total of 6 valence electrons, with two bonds to oxygen atoms and two bonds to chlorine atoms. A hydrogen atom can form a maximum of one covalent bond. Carbon and hydrogen do not typically form hydrogen bonds.
Sulfur can form up to two covalent bonds because it has six. The sulfur atom has a total of 6 valence electrons, with two bonds to oxygen atoms and two bonds to chlorine atoms. A hydrogen atom can form a maximum of one covalent bond. Carbon and hydrogen do not typically form hydrogen bonds. If sulfur has an atomic number of 16 how many covalent bonds does it form?
Oxygen Diagram Atom Ossigeno Atomic Struttura Atomica
If sulfur has an atomic number of 16 how many covalent bonds does it form? The sulfur atom has a total of 6 valence electrons, with two bonds to oxygen atoms and two bonds to chlorine atoms. Carbon and hydrogen do not typically form hydrogen bonds. Sulfur can form up to two covalent bonds because it has six. A hydrogen.
Carbon Oxygen Covalent Bond vrogue.co
A hydrogen atom can form a maximum of one covalent bond. Carbon and hydrogen do not typically form hydrogen bonds. The sulfur atom has a total of 6 valence electrons, with two bonds to oxygen atoms and two bonds to chlorine atoms. Sulfur can form up to two covalent bonds because it has six. If sulfur has an atomic number.
PPT Chemistry Bonding PowerPoint Presentation, free download ID
The sulfur atom has a total of 6 valence electrons, with two bonds to oxygen atoms and two bonds to chlorine atoms. If sulfur has an atomic number of 16 how many covalent bonds does it form? A hydrogen atom can form a maximum of one covalent bond. Carbon and hydrogen do not typically form hydrogen bonds. Sulfur can form.
Basic Cell Biology
The sulfur atom has a total of 6 valence electrons, with two bonds to oxygen atoms and two bonds to chlorine atoms. If sulfur has an atomic number of 16 how many covalent bonds does it form? Sulfur can form up to two covalent bonds because it has six. Carbon and hydrogen do not typically form hydrogen bonds. A hydrogen.
A guide for A level students KNOCKHARDY PUBLISHING ppt download
Sulfur can form up to two covalent bonds because it has six. Carbon and hydrogen do not typically form hydrogen bonds. A hydrogen atom can form a maximum of one covalent bond. If sulfur has an atomic number of 16 how many covalent bonds does it form? The sulfur atom has a total of 6 valence electrons, with two bonds.
Atoms, Molecules, and Life ppt download
Carbon and hydrogen do not typically form hydrogen bonds. If sulfur has an atomic number of 16 how many covalent bonds does it form? A hydrogen atom can form a maximum of one covalent bond. Sulfur can form up to two covalent bonds because it has six. The sulfur atom has a total of 6 valence electrons, with two bonds.
PPT Remembering General Chemistry Electronic Structure and Bonding
A hydrogen atom can form a maximum of one covalent bond. If sulfur has an atomic number of 16 how many covalent bonds does it form? Sulfur can form up to two covalent bonds because it has six. Carbon and hydrogen do not typically form hydrogen bonds. The sulfur atom has a total of 6 valence electrons, with two bonds.
Notes Covalent Bonds 9/24/18 ppt download
Sulfur can form up to two covalent bonds because it has six. A hydrogen atom can form a maximum of one covalent bond. The sulfur atom has a total of 6 valence electrons, with two bonds to oxygen atoms and two bonds to chlorine atoms. Carbon and hydrogen do not typically form hydrogen bonds. If sulfur has an atomic number.
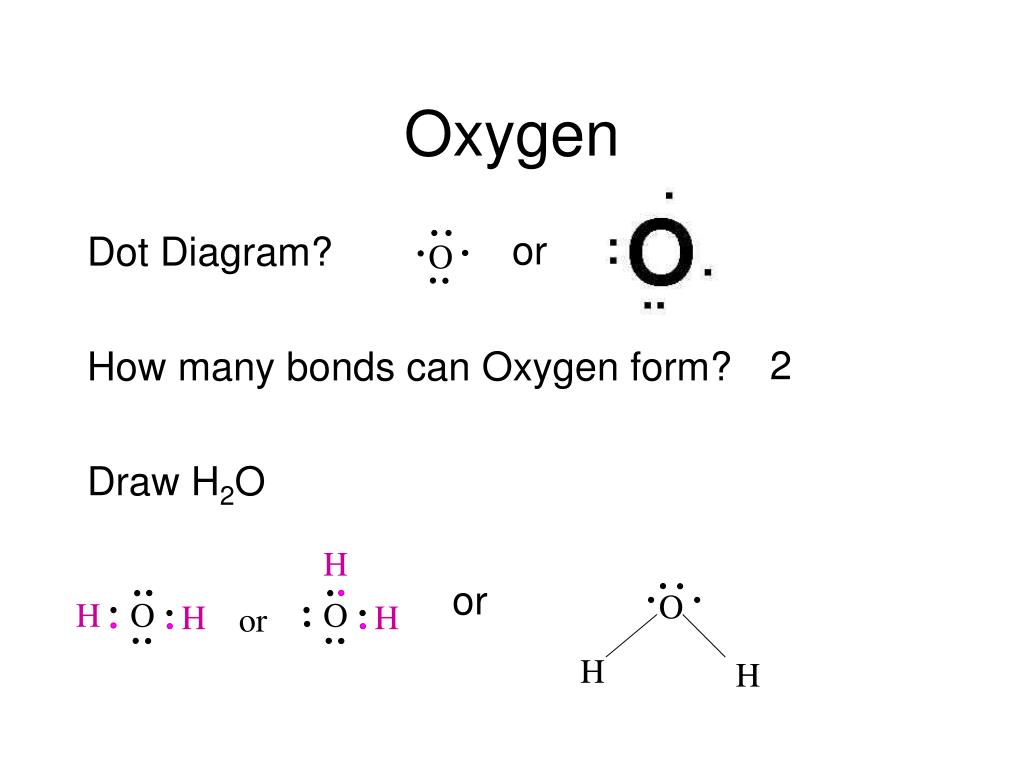
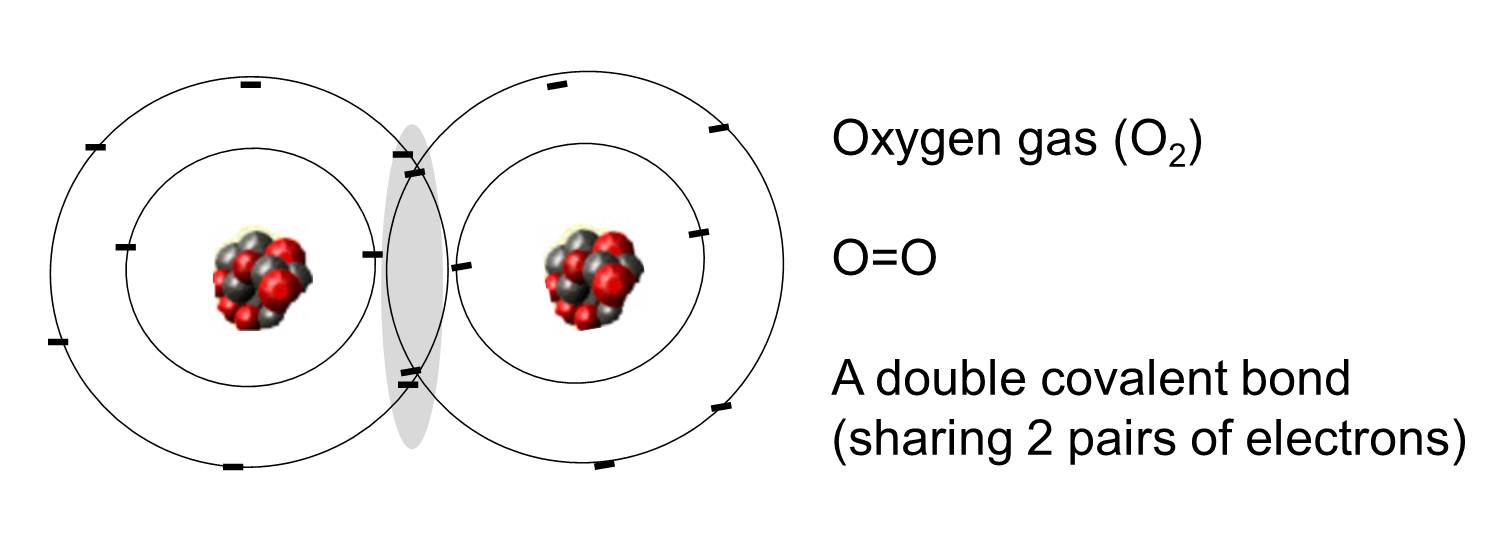
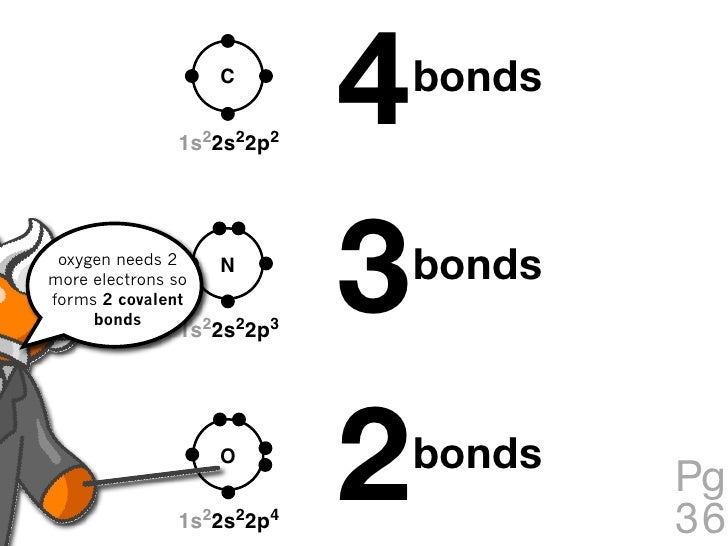
C 1s22s22p2 4 bonds oxygen
Carbon and hydrogen do not typically form hydrogen bonds. A hydrogen atom can form a maximum of one covalent bond. The sulfur atom has a total of 6 valence electrons, with two bonds to oxygen atoms and two bonds to chlorine atoms. Sulfur can form up to two covalent bonds because it has six. If sulfur has an atomic number.
Covalent Bonding GCSE Chemistry Revision
Sulfur can form up to two covalent bonds because it has six. A hydrogen atom can form a maximum of one covalent bond. The sulfur atom has a total of 6 valence electrons, with two bonds to oxygen atoms and two bonds to chlorine atoms. Carbon and hydrogen do not typically form hydrogen bonds. If sulfur has an atomic number.
The Sulfur Atom Has A Total Of 6 Valence Electrons, With Two Bonds To Oxygen Atoms And Two Bonds To Chlorine Atoms.
A hydrogen atom can form a maximum of one covalent bond. Carbon and hydrogen do not typically form hydrogen bonds. If sulfur has an atomic number of 16 how many covalent bonds does it form? Sulfur can form up to two covalent bonds because it has six.