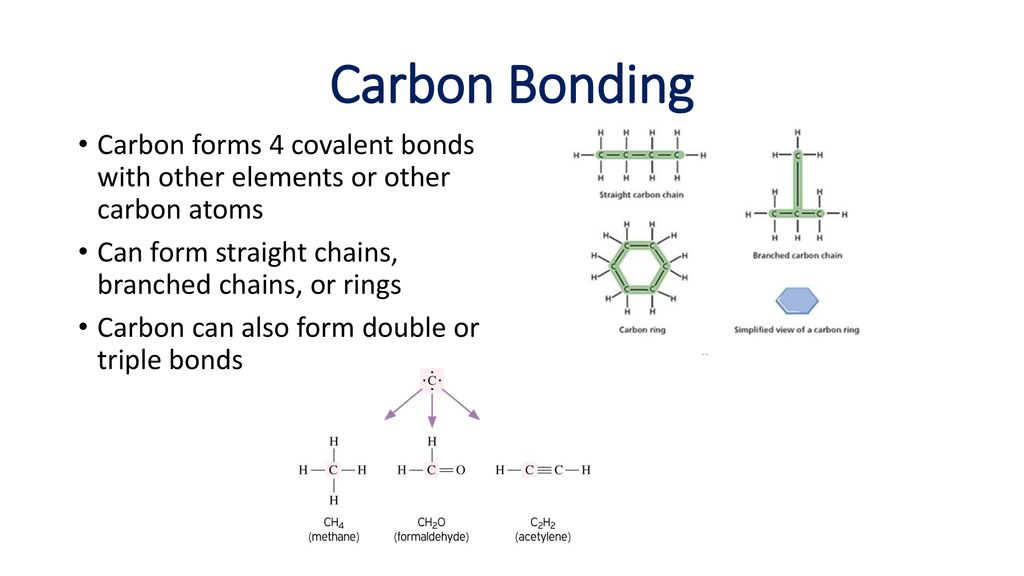
How Many Bonds Are Typically Formed By Carbon - This allows it to create a diverse range of organic. The simplest carbon molecule is methane (ch 4), depicted here. How many bonds does carbon form? Study with quizlet and memorize flashcards containing terms like how many bonds does carbon form?, what can carbon compounds form?, what. Carbon typically forms four covalent bonds in compounds due to its tetravalent nature. Carbon can form four covalent bonds to create an organic molecule. This is because it has four valence electrons and needs four more to fill its. Each bond allows carbon to share. Carbon needs 4 more electrons to complete its octet, which it achieves by forming covalent bonds. Carbon typically forms four bonds.
Carbon typically forms four bonds. This allows it to create a diverse range of organic. Carbon typically forms four covalent bonds in compounds due to its tetravalent nature. How many bonds does carbon form? The simplest carbon molecule is methane (ch 4), depicted here. This is because it has four valence electrons and needs four more to fill its. Each bond allows carbon to share. Carbon can form four covalent bonds to create an organic molecule. Study with quizlet and memorize flashcards containing terms like how many bonds does carbon form?, what can carbon compounds form?, what. Carbon needs 4 more electrons to complete its octet, which it achieves by forming covalent bonds.
Carbon typically forms four bonds. How many bonds does carbon form? The simplest carbon molecule is methane (ch 4), depicted here. This is because it has four valence electrons and needs four more to fill its. Carbon can form four covalent bonds to create an organic molecule. Carbon needs 4 more electrons to complete its octet, which it achieves by forming covalent bonds. Study with quizlet and memorize flashcards containing terms like how many bonds does carbon form?, what can carbon compounds form?, what. Each bond allows carbon to share. Carbon typically forms four covalent bonds in compounds due to its tetravalent nature. This allows it to create a diverse range of organic.
Carbon Compounds. ppt download
Each bond allows carbon to share. This allows it to create a diverse range of organic. Carbon typically forms four bonds. Carbon typically forms four covalent bonds in compounds due to its tetravalent nature. This is because it has four valence electrons and needs four more to fill its.
Organic Chemistry Chapter ppt download
The simplest carbon molecule is methane (ch 4), depicted here. Carbon needs 4 more electrons to complete its octet, which it achieves by forming covalent bonds. This is because it has four valence electrons and needs four more to fill its. Carbon typically forms four covalent bonds in compounds due to its tetravalent nature. Carbon can form four covalent bonds.
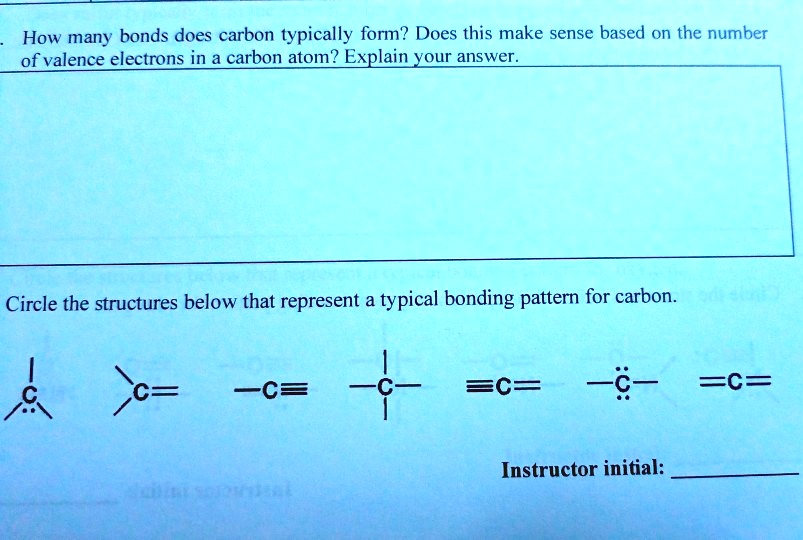
SOLVED How many bonds does carbon typically form? Does this make sense
This allows it to create a diverse range of organic. Carbon can form four covalent bonds to create an organic molecule. Carbon typically forms four bonds. Carbon needs 4 more electrons to complete its octet, which it achieves by forming covalent bonds. Carbon typically forms four covalent bonds in compounds due to its tetravalent nature.
1 How many bonds does carbon typically form? Does
This allows it to create a diverse range of organic. This is because it has four valence electrons and needs four more to fill its. Study with quizlet and memorize flashcards containing terms like how many bonds does carbon form?, what can carbon compounds form?, what. Carbon can form four covalent bonds to create an organic molecule. The simplest carbon.
The Chemistry of Carbon = Chemistry of Life ppt download
Carbon typically forms four bonds. This allows it to create a diverse range of organic. Carbon typically forms four covalent bonds in compounds due to its tetravalent nature. Study with quizlet and memorize flashcards containing terms like how many bonds does carbon form?, what can carbon compounds form?, what. How many bonds does carbon form?
Unit 2 (Biochemistry) Notes, Part 1 Atomic And Molecular Structure
Carbon typically forms four bonds. This allows it to create a diverse range of organic. This is because it has four valence electrons and needs four more to fill its. Each bond allows carbon to share. Study with quizlet and memorize flashcards containing terms like how many bonds does carbon form?, what can carbon compounds form?, what.
Organic Molecules Chapter ppt download
Carbon can form four covalent bonds to create an organic molecule. Carbon needs 4 more electrons to complete its octet, which it achieves by forming covalent bonds. Study with quizlet and memorize flashcards containing terms like how many bonds does carbon form?, what can carbon compounds form?, what. How many bonds does carbon form? Carbon typically forms four covalent bonds.
How Many Forms Does Carbon Have at Claire Mcvicars blog
This allows it to create a diverse range of organic. This is because it has four valence electrons and needs four more to fill its. Study with quizlet and memorize flashcards containing terms like how many bonds does carbon form?, what can carbon compounds form?, what. Carbon needs 4 more electrons to complete its octet, which it achieves by forming.
MACROMOLECULES a.k.a. BioMolecules a.k.a. Organic Molecules ppt download
The simplest carbon molecule is methane (ch 4), depicted here. This allows it to create a diverse range of organic. Carbon can form four covalent bonds to create an organic molecule. Carbon needs 4 more electrons to complete its octet, which it achieves by forming covalent bonds. How many bonds does carbon form?
Chapter 9 Carbon & Its Compounds. ppt video online download
Each bond allows carbon to share. Carbon can form four covalent bonds to create an organic molecule. Carbon typically forms four bonds. Carbon typically forms four covalent bonds in compounds due to its tetravalent nature. The simplest carbon molecule is methane (ch 4), depicted here.
How Many Bonds Does Carbon Form?
The simplest carbon molecule is methane (ch 4), depicted here. This allows it to create a diverse range of organic. Carbon typically forms four bonds. This is because it has four valence electrons and needs four more to fill its.
Carbon Needs 4 More Electrons To Complete Its Octet, Which It Achieves By Forming Covalent Bonds.
Carbon can form four covalent bonds to create an organic molecule. Carbon typically forms four covalent bonds in compounds due to its tetravalent nature. Study with quizlet and memorize flashcards containing terms like how many bonds does carbon form?, what can carbon compounds form?, what. Each bond allows carbon to share.