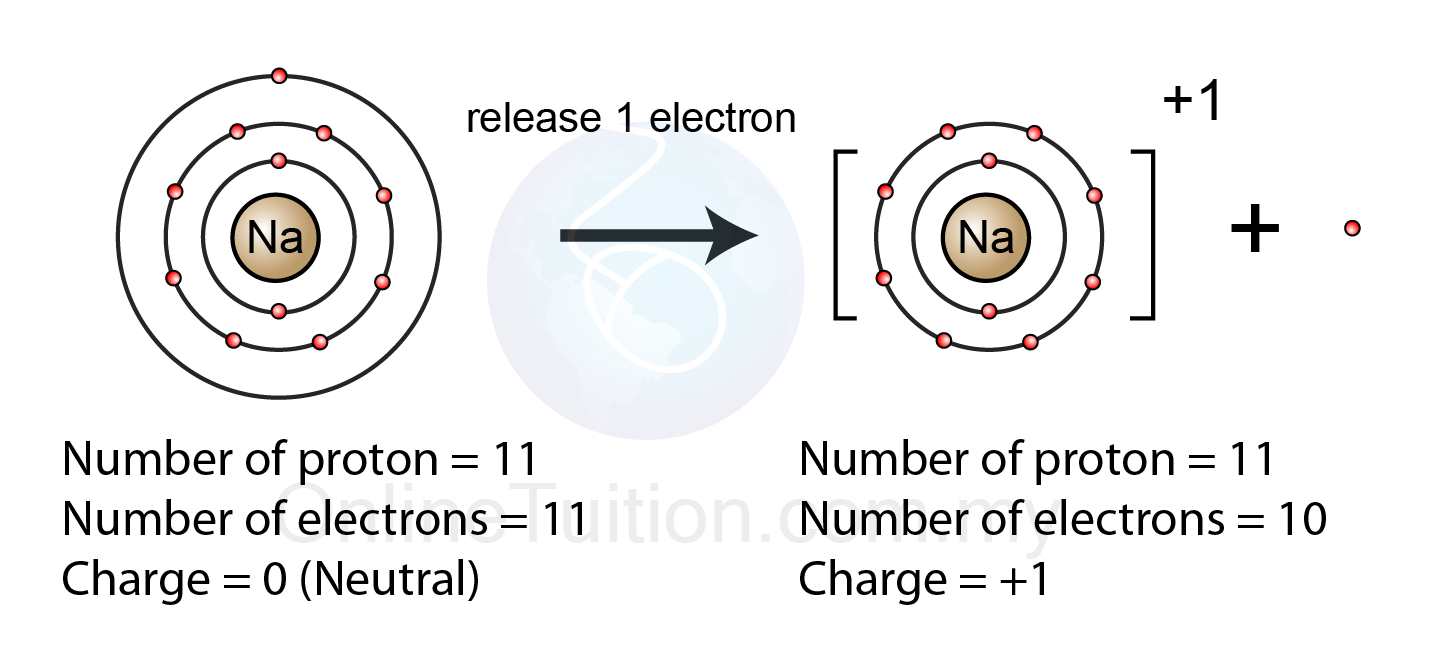
How Do Cations Form - Cations are positive ions, so an atom is supposed to lose electrons to become a cation. If an element has less than four valence. However, when elements lose electrons to become stable, they are called cations. Alkali metals generally form cations by losing their outermost electron to achieve a stable electron configuration. Atoms in groups 1, 2, and 13 tend to lose electrons to form positively charged cations, while atoms in groups 15, 16, and 17 tend to gain. Anions are formed when an atom gains electrons. When metals form ions, they tend to do so by losing their electrons to gain a positive charge.
Alkali metals generally form cations by losing their outermost electron to achieve a stable electron configuration. Atoms in groups 1, 2, and 13 tend to lose electrons to form positively charged cations, while atoms in groups 15, 16, and 17 tend to gain. When metals form ions, they tend to do so by losing their electrons to gain a positive charge. However, when elements lose electrons to become stable, they are called cations. If an element has less than four valence. Anions are formed when an atom gains electrons. Cations are positive ions, so an atom is supposed to lose electrons to become a cation.
If an element has less than four valence. Atoms in groups 1, 2, and 13 tend to lose electrons to form positively charged cations, while atoms in groups 15, 16, and 17 tend to gain. However, when elements lose electrons to become stable, they are called cations. Anions are formed when an atom gains electrons. Cations are positive ions, so an atom is supposed to lose electrons to become a cation. When metals form ions, they tend to do so by losing their electrons to gain a positive charge. Alkali metals generally form cations by losing their outermost electron to achieve a stable electron configuration.
Chemistry ppt download
When metals form ions, they tend to do so by losing their electrons to gain a positive charge. Cations are positive ions, so an atom is supposed to lose electrons to become a cation. Anions are formed when an atom gains electrons. Alkali metals generally form cations by losing their outermost electron to achieve a stable electron configuration. However, when.
Cations and Anions Definitions, Examples, and Differences
When metals form ions, they tend to do so by losing their electrons to gain a positive charge. Atoms in groups 1, 2, and 13 tend to lose electrons to form positively charged cations, while atoms in groups 15, 16, and 17 tend to gain. Cations are positive ions, so an atom is supposed to lose electrons to become a.
Forming Ions GCSE Chemistry Revision
Anions are formed when an atom gains electrons. Alkali metals generally form cations by losing their outermost electron to achieve a stable electron configuration. However, when elements lose electrons to become stable, they are called cations. Cations are positive ions, so an atom is supposed to lose electrons to become a cation. When metals form ions, they tend to do.
Cation vs Anion Definitions & Periodic Table Guide Technology Networks
When metals form ions, they tend to do so by losing their electrons to gain a positive charge. Atoms in groups 1, 2, and 13 tend to lose electrons to form positively charged cations, while atoms in groups 15, 16, and 17 tend to gain. However, when elements lose electrons to become stable, they are called cations. Alkali metals generally.
What Is A Ion Cations at Annie Miller blog
However, when elements lose electrons to become stable, they are called cations. Anions are formed when an atom gains electrons. When metals form ions, they tend to do so by losing their electrons to gain a positive charge. If an element has less than four valence. Atoms in groups 1, 2, and 13 tend to lose electrons to form positively.
Structure & Bonding. ppt download
Alkali metals generally form cations by losing their outermost electron to achieve a stable electron configuration. If an element has less than four valence. Atoms in groups 1, 2, and 13 tend to lose electrons to form positively charged cations, while atoms in groups 15, 16, and 17 tend to gain. However, when elements lose electrons to become stable, they.
Valence elecrtrons and chemical properties ppt download
Atoms in groups 1, 2, and 13 tend to lose electrons to form positively charged cations, while atoms in groups 15, 16, and 17 tend to gain. If an element has less than four valence. When metals form ions, they tend to do so by losing their electrons to gain a positive charge. Alkali metals generally form cations by losing.
Ions. ppt download
If an element has less than four valence. Anions are formed when an atom gains electrons. Cations are positive ions, so an atom is supposed to lose electrons to become a cation. However, when elements lose electrons to become stable, they are called cations. Alkali metals generally form cations by losing their outermost electron to achieve a stable electron configuration.
Unit 5 Ionic Bonding & Nomenclature ppt download
Anions are formed when an atom gains electrons. If an element has less than four valence. Atoms in groups 1, 2, and 13 tend to lose electrons to form positively charged cations, while atoms in groups 15, 16, and 17 tend to gain. Cations are positive ions, so an atom is supposed to lose electrons to become a cation. Alkali.
Atoms and Ions Pg ppt download
When metals form ions, they tend to do so by losing their electrons to gain a positive charge. Cations are positive ions, so an atom is supposed to lose electrons to become a cation. However, when elements lose electrons to become stable, they are called cations. If an element has less than four valence. Anions are formed when an atom.
However, When Elements Lose Electrons To Become Stable, They Are Called Cations.
If an element has less than four valence. Anions are formed when an atom gains electrons. Cations are positive ions, so an atom is supposed to lose electrons to become a cation. Atoms in groups 1, 2, and 13 tend to lose electrons to form positively charged cations, while atoms in groups 15, 16, and 17 tend to gain.
Alkali Metals Generally Form Cations By Losing Their Outermost Electron To Achieve A Stable Electron Configuration.
When metals form ions, they tend to do so by losing their electrons to gain a positive charge.








+form+when+an+atom+has+more+protons+than+electrons..jpg)