Compounds That React With Acids To Form Salts - Lets take the ionic formula for calcium chloride, cacl_2. How do ionic compounds dissolve? In living things, organic compounds are often called biomolecules, but. Compounds that contain carbon, hydrogen, and oxygen are organic. Is it possible to distinguish between ionic and. Calcium is an alkaline earth metal in the second column of the periodic table. Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Of course, not all ionic compounds are soluble in. How do ionic compounds conduct electricity in water? Structural isomers are compounds of the same chemical formula but different connectivities.
Compounds that contain carbon, hydrogen, and oxygen are organic. Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Lets take the ionic formula for calcium chloride, cacl_2. Structural isomers are compounds of the same chemical formula but different connectivities. How do ionic compounds dissolve? Is it possible to distinguish between ionic and. Calcium is an alkaline earth metal in the second column of the periodic table. In living things, organic compounds are often called biomolecules, but. How do ionic compounds conduct electricity in water? Of course, not all ionic compounds are soluble in.
How do ionic compounds conduct electricity in water? In living things, organic compounds are often called biomolecules, but. Of course, not all ionic compounds are soluble in. How do ionic compounds dissolve? Is it possible to distinguish between ionic and. Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Calcium is an alkaline earth metal in the second column of the periodic table. Compounds that contain carbon, hydrogen, and oxygen are organic. Structural isomers are compounds of the same chemical formula but different connectivities. Lets take the ionic formula for calcium chloride, cacl_2.
Salt And Water Reaction
Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Structural isomers are compounds of the same chemical formula but different connectivities. Lets take the ionic formula for calcium chloride, cacl_2. In living things, organic compounds are often called biomolecules, but. Of course, not all ionic compounds are soluble in.
Question Video Identifying the Acid Required to Make a Specific Salt
Compounds that contain carbon, hydrogen, and oxygen are organic. Of course, not all ionic compounds are soluble in. Lets take the ionic formula for calcium chloride, cacl_2. Calcium is an alkaline earth metal in the second column of the periodic table. Is it possible to distinguish between ionic and.
Chapter 14 Acids, Bases, and pH ppt download
Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Calcium is an alkaline earth metal in the second column of the periodic table. In living things, organic compounds are often called biomolecules, but. Of course, not all ionic compounds are soluble in. Is it possible to distinguish between ionic and.
Reaction of Metals and NonMetals with Acids Teachoo Concepts
Calcium is an alkaline earth metal in the second column of the periodic table. Of course, not all ionic compounds are soluble in. How do ionic compounds conduct electricity in water? In living things, organic compounds are often called biomolecules, but. Lets take the ionic formula for calcium chloride, cacl_2.
Chapter 5 Compounds are classified in different ways ppt video online
In living things, organic compounds are often called biomolecules, but. How do ionic compounds conduct electricity in water? Calcium is an alkaline earth metal in the second column of the periodic table. Of course, not all ionic compounds are soluble in. Lets take the ionic formula for calcium chloride, cacl_2.
5.2 Salts. ppt download
Is it possible to distinguish between ionic and. Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Lets take the ionic formula for calcium chloride, cacl_2. Compounds that contain carbon, hydrogen, and oxygen are organic. Of course, not all ionic compounds are soluble in.
5.1 Acids and Bases. ppt download
In living things, organic compounds are often called biomolecules, but. Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. How do ionic compounds dissolve? Is it possible to distinguish between ionic and. Of course, not all ionic compounds are soluble in.
Types of Acidic, Basic and Neutral Salts Poster
In living things, organic compounds are often called biomolecules, but. Calcium is an alkaline earth metal in the second column of the periodic table. Is it possible to distinguish between ionic and. How do ionic compounds dissolve? How do ionic compounds conduct electricity in water?
Compounds. ppt video online download
Structural isomers are compounds of the same chemical formula but different connectivities. Is it possible to distinguish between ionic and. In living things, organic compounds are often called biomolecules, but. Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Lets take the ionic formula for calcium chloride, cacl_2.
Reactions of Acids. ppt download
Of course, not all ionic compounds are soluble in. How do ionic compounds dissolve? Because water is a solvent that is capable of solvating cations, and to a lesser extents anions. Calcium is an alkaline earth metal in the second column of the periodic table. How do ionic compounds conduct electricity in water?
Calcium Is An Alkaline Earth Metal In The Second Column Of The Periodic Table.
In living things, organic compounds are often called biomolecules, but. Lets take the ionic formula for calcium chloride, cacl_2. How do ionic compounds dissolve? Is it possible to distinguish between ionic and.
Structural Isomers Are Compounds Of The Same Chemical Formula But Different Connectivities.
Compounds that contain carbon, hydrogen, and oxygen are organic. Of course, not all ionic compounds are soluble in. How do ionic compounds conduct electricity in water? Because water is a solvent that is capable of solvating cations, and to a lesser extents anions.
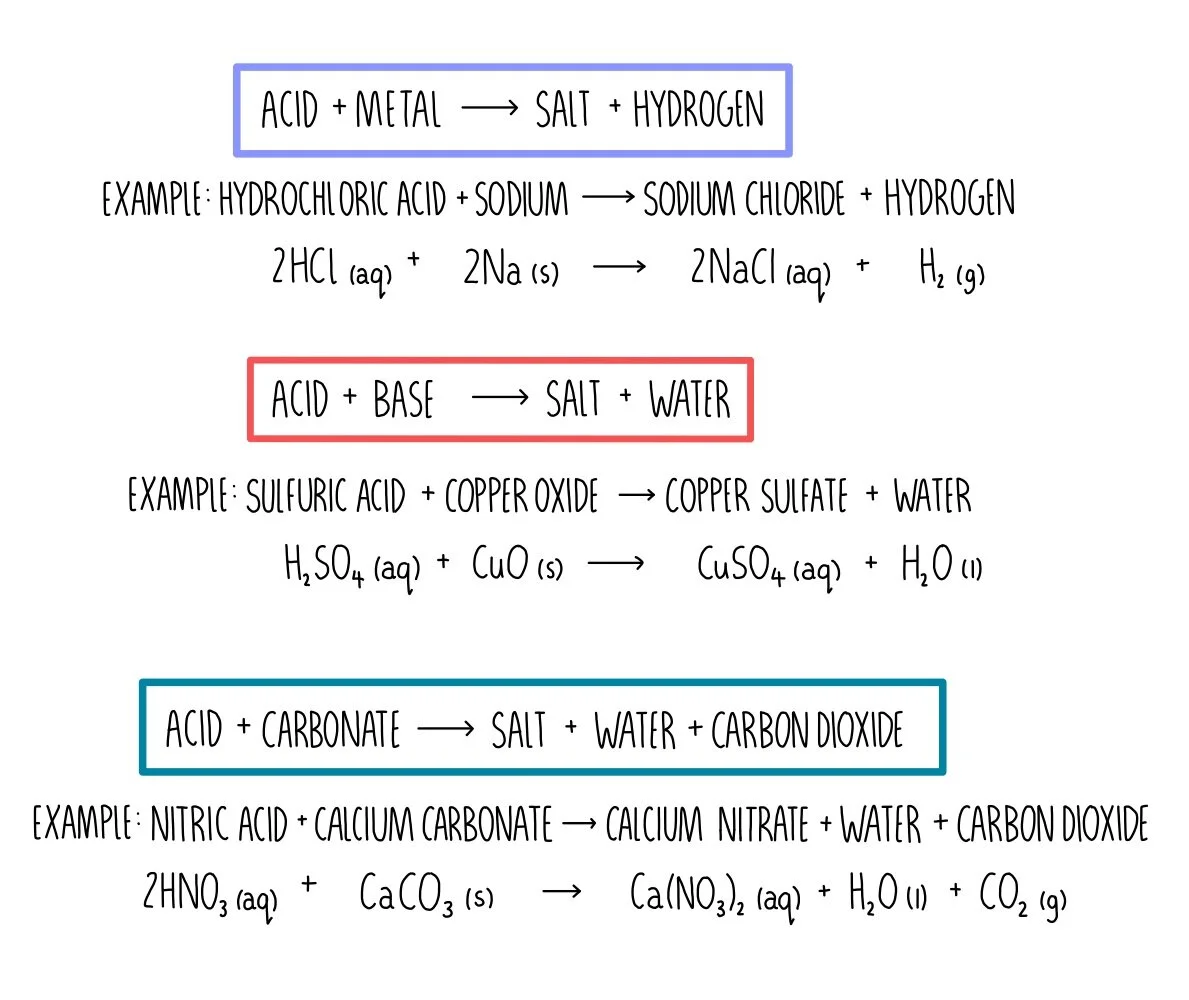

+Acids+react+with+bases+to+produce+salts+and+water..jpg)






