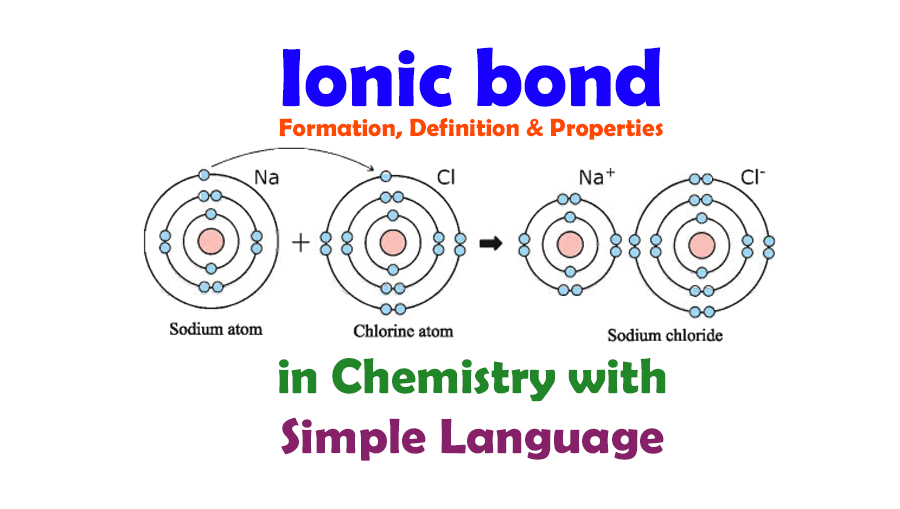
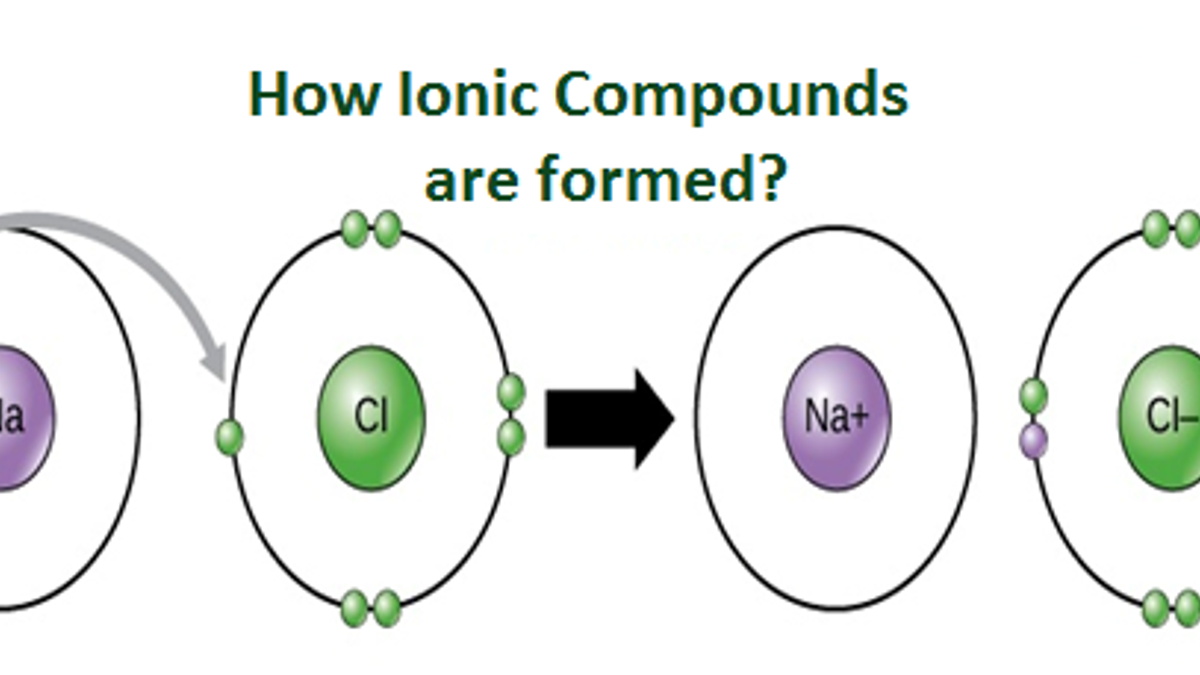
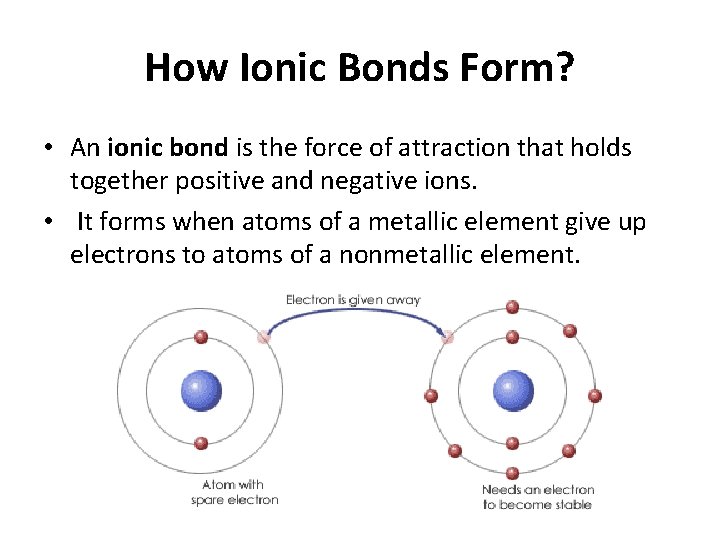
An Ionic Bond Is Formed By - In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. What is an ionic bond? An ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between. Ionic bonding can result from a redox reaction when atoms of an element (usually metal), whose ionization energy is low, give some of their. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Atoms or ions with large electronegativity differences form ionic bonds. Those with small or no electronegativity.
Ionic bonding can result from a redox reaction when atoms of an element (usually metal), whose ionization energy is low, give some of their. In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. Those with small or no electronegativity. An ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between. Atoms or ions with large electronegativity differences form ionic bonds. What is an ionic bond? Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound.
Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. An ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between. Ionic bonding can result from a redox reaction when atoms of an element (usually metal), whose ionization energy is low, give some of their. In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. What is an ionic bond? Atoms or ions with large electronegativity differences form ionic bonds. Those with small or no electronegativity.
Examples of Ionic Bonds and Compounds
What is an ionic bond? An ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between. In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. Those with small or no electronegativity. Ionic bonding can result from a.
Basic Chemistry Chapter ppt download
Atoms or ions with large electronegativity differences form ionic bonds. What is an ionic bond? Ionic bonding can result from a redox reaction when atoms of an element (usually metal), whose ionization energy is low, give some of their. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Those with small.
Ionic Bond and Ionic Bond Formation, Definition, Properties in
Those with small or no electronegativity. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. What is an ionic bond? An ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between. Atoms or ions with large electronegativity differences form ionic.
ionic bond Definition, Properties, Examples, & Facts Britannica
In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. An ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between. Atoms or ions with large electronegativity differences form ionic bonds. Those with small or no electronegativity. Ionic.
Physical science Chapter 20, Section 1 ppt download
Those with small or no electronegativity. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. What is an ionic bond? Atoms or ions with large electronegativity differences form ionic bonds. Ionic bonding can result from a redox reaction when atoms of an element (usually metal), whose ionization energy is low, give.
Ionic Bond Formation Edexcel GCSE Chemistry Revision
What is an ionic bond? Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Ionic bonding can result from a redox reaction when atoms of an element (usually metal), whose ionization energy is low, give some of their. Atoms or ions with large electronegativity differences form ionic bonds. In this lesson,.
What are Ionic Compounds and how they are formed?
Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Ionic bonding can result from a redox reaction when atoms of an element (usually metal), whose ionization energy is low, give some of their. What is an ionic bond? Those with small or no electronegativity. In this lesson, you'll discover how metals.
Ionic Bond Definition and Examples
An ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between. Ionic bonding can result from a redox reaction when atoms of an element (usually metal), whose ionization energy is low, give some of their. Atoms or ions with large electronegativity differences form ionic bonds. In this lesson, you'll.
Ionic Bonds Essential Question How do ionic bonds
Those with small or no electronegativity. Atoms or ions with large electronegativity differences form ionic bonds. What is an ionic bond? Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Ionic bonding can result from a redox reaction when atoms of an element (usually metal), whose ionization energy is low, give.
Chapter 21 The Nature of Matter ppt download
In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. What is an ionic bond? Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Atoms or ions with large electronegativity differences form ionic bonds. Those with small or no electronegativity.
Ionic Bonding Can Result From A Redox Reaction When Atoms Of An Element (Usually Metal), Whose Ionization Energy Is Low, Give Some Of Their.
What is an ionic bond? In this lesson, you'll discover how metals transfer electrons to nonmetals to form oppositely charged ions, and how these electrostatic attractions. Those with small or no electronegativity. An ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between.
Atoms Or Ions With Large Electronegativity Differences Form Ionic Bonds.
Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound.